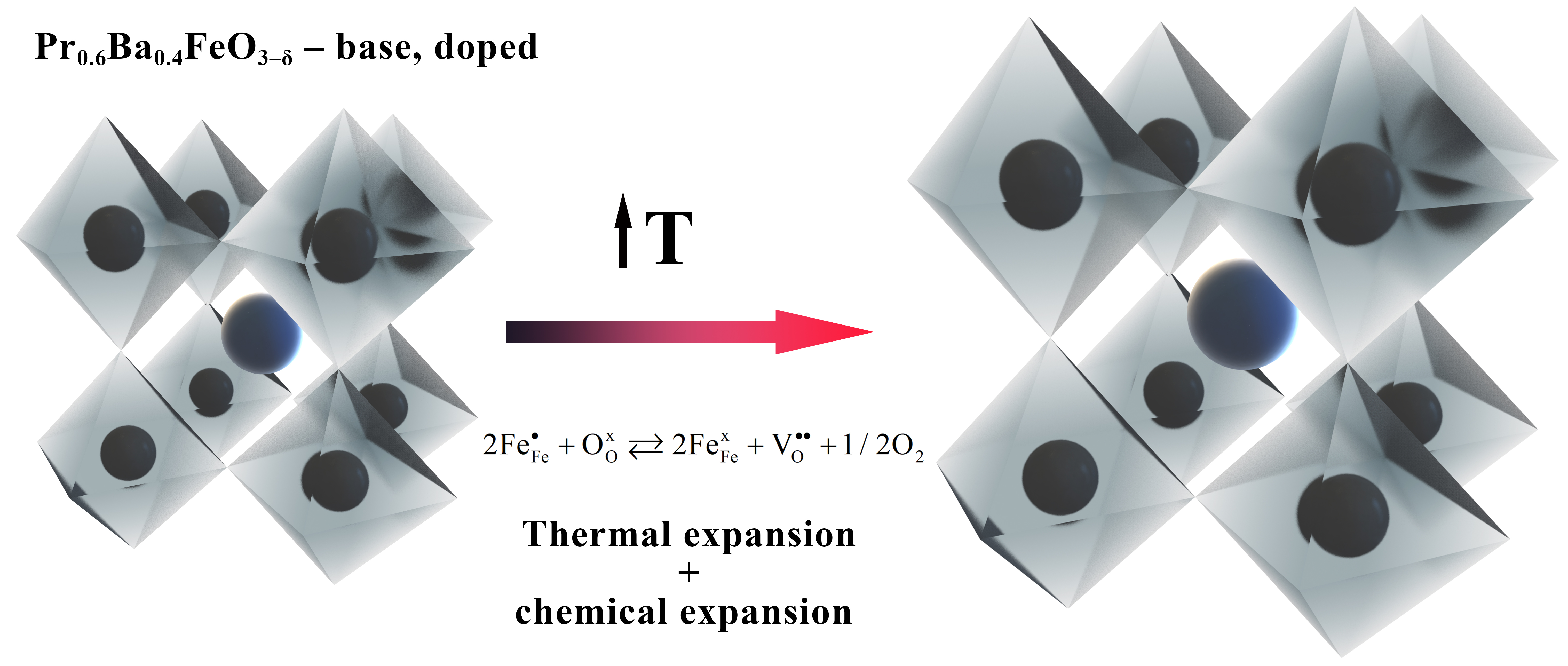
Thermomechanical behavior of promising mixed ion-electron conductors based on co-doped barium ferrite
Abstract
Keywords
Full Text:
PDFReferences
Le TT, Sharma P, Bora BJ, Tran VD, et al., Fueling the future: A comprehensive review of hydrogen energy systems and their challenges, Int. J. Hydrogen Energy., 54 (2024) 791–816. https://doi.org/10.1016/j.ijhydene.2023.08.044
Hassan Q, Viktor P, J. Al-Musawi T, Mahmood AB, et al., The renewable energy role in the global energy Transformations, Renewable Energy Focus, 48 (2024) 100545. https://doi.org/10.1016/j.ref.2024.100545
Zhang L, Jia C, Bai F, Wang W, et al., A comprehensive review of the promising clean energy carrier: Hydrogen production, transportation, storage, and utilization (HPTSU) technologies, Fuel, 355 (2024) 129455. https://doi.org/10.1016/j.fuel.2023.129455
Tariq AH, Kazmi SAA, Hassan M, Muhammed Ali SA, Anwar M, Analysis of fuel cell integration with hybrid microgrid systems for clean energy: A comparative review, Int. J. Hydrogen Energy, 52 (2024) 1005–34. https://doi.org/10.1016/j.ijhydene.2023.07.238
Xu Y, Cai S, Chi B, Tu Z, Technological limitations and recent developments in a solid oxide electrolyzer cell: A review, Int. J. Hydrogen Energy, 50 (2024) 548–91. https://doi.org/10.1016/j.ijhydene.2023.08.314
Zarabi Golkhatmi S, Asghar MI, Lund PD, A review on solid oxide fuel cell durability: Latest progress, mechanisms, and study tools, Renew. Sustain. Energy Rev., 161 (2022) 112339. https://doi.org/10.1016/j.rser.2022.112339
Ndubuisi A, Abouali S, Singh K, Thangadurai V, Recent advances, practical challenges, and perspectives of intermediate temperature solid oxide fuel cell cathodes, J. Mater. Chem. A, 10 (2022) 2196–227. https://doi.org/10.1039/D1TA08475E
Tarutin AP, Filonova EA, Ricote S, Medvedev DA, Shao Z, Chemical design of oxygen electrodes for solid oxide electrochemical cells: A guide, Sustainable Energy Technologies and Assessments, 57 (2023) 103185. https://doi.org/10.1016/j.seta.2023.103185
Filonova E, Pikalova E, Overview of approaches to increase the electrochemical activity of conventional perovskite air electrodes, Materials (Basel), 16(14) (2023) 4967. https://doi.org/10.3390/ma16144967
Han N, Shen Z, Zhao X, Chen R, Thakur VK, Perovskite oxides for oxygen transport: Chemistry and material horizons, Sci. Total Environ., 806 (2022) 151213. https://doi.org/10.1016/j.scitotenv.2021.151213
Chen G, Feldhoff A, Weidenkaff A, Li C, et al., Roadmap for sustainable mixed ionic‐electronic conducting membranes, Adv. Funct. Mater., 32(6) (2022) 2105702. https://doi.org/10.1002/adfm.202105702
Ni C, Zhou J, Zhang Z, Li S, et al., Iron-based electrode materials for solid oxide fuel cells and electrolysers, Energy Environ. Sci., 14 (2021) 6287–319. https://doi.org/10.1039/D1EE01420J
Gordeeva MA, Starostina IA, Murashkina AA, Vdovin GK, Medvedev DA, Transport properties and phase stability of Ni-doped Pr0.6Ba0.4FeO3–δ as potential symmetrical electrodes for proton-conducting electrochemical cells, Int. J. Hydrogen Energy, 91 (2024) 16–28. https://doi.org/10.1016/j.ijhydene.2024.10.115
Gordeeva MA, Tarutin AP, Vdovin GK, Matkin DE, et al., Ba-containing ferrites, Pr1–xBaxFeO3–δ, as symmetrical electrodes and their functional properties in both oxidizing and reducing atmospheres, Ceram. Int., 50(20) (2024) 40417–40428. https://doi.org/10.1016/j.ceramint.2024.02.067
Kharton VV, Yaremchenko AA, Patrakeev MV, Naumovich EN, Marques FMB, Thermal and chemical induced expansion of La0.3Sr0.7(Fe,Ga)O3–δ ceramics, J. Eur. Ceram. Soc., 23 (2003) 1417–26. https://doi.org/10.1016/S0955-2219(02)00308-4
Kostogloudis G, Structural, thermal and electrical properties of Pr0.5Sr0.5Co1−yNiyO3–δ perovskite-type oxides, Solid State Ion., 109(1–2) (1998) 43–53. https://doi.org/10.1016/S0167-2738(98)00007-1
DOI: https://doi.org/10.15826/elmattech.2025.4.048
Copyright (c) 2025 Maria A. Gordeeva, Liana R. Tarutina, Anna A. Murashkina

This work is licensed under a Creative Commons Attribution 4.0 International License.

