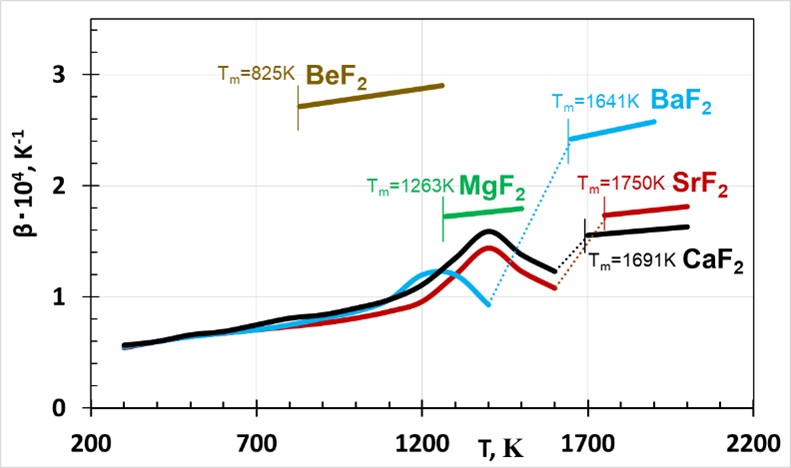
Thermal expansion of alkali and alkaline earth halides in solid and molten states
Abstract
Keywords
Full Text:
PDFReferences
Tiwari R, Devendra K, Dipendra KV, Parwati K, et al., Fundamental chemical and physical properties of electrolytes in energy storage devices: A review, J. Energy Storage., 81 (2024) 110361. https://doi.org/10.1016/j.est.2023.110361
Bo C, Phase change behaviour of some latent heat storage media based on calcium chloride hexahydrate, Solar Energy, 83(4) (2009) 485–500. https://doi.org/10.1016/j.solener.2008.09.004
Tao B, Zhong D, Li H, Wang G, et al., Halide solid-state electrolytes for all-solid-state batteries: structural design, synthesis, environmental stability, interface optimization and challenges, Chem. Sci., 14 (2023) 8693–8722. https://doi.org/10.1039/d3sc02093b
Babaev BD, Principles of heat accumulation and heat accumulating materials in use, High Temperature, 52(5) (2014) 736–751. https://doi.org/10.1134/S0018151X14050010
Redkin A, Apisarov A, Dedyukhin A, Kovrov V, et al., Recent developments in low-temperature electrolysis of aluminum, ECS Transactions, 50(11) (2012) 205–213. https://doi.org/10.1149/05011.0205ecst
Zaikov Yu, Batukhtin V, Shurov N, Suzdaltsev A, High-temperature electrochemistry of calcium, Electrochem. Mater. Technol., 1 (2022) 20221007. https://doi.org/10.15826/elmattech.2022.1.007
Haarberg GM. Alkali and Alkaline Earth Metal Production by Molten Salt. In: Kreysa G, Ota K, Savinell RF. (eds) Encyclopedia of Applied Electrochemistry. New York, United States: Springer; 2014. p. 21–25. https://doi.org/10.1007/978-1-4419-6996-5_451
Wang Z, Cheng Y, He F, Lv Z, Electroplating of refractory metals in molten salts: A Review, JOM, 76(8) (2024) 4050–4067. https://doi.org/10.1007/s11837-024-06695-z
Fache M, Voigt L, Colle JY, Hald J, et al., Thermophysical properties of FUNaK (NaF-KF-UF4) eutectics, Materials, 17(11) (2024) 2776. https://doi.org/10.3390/ma17112776
Magnusson J, Memmott M, Munro T, Review of thermophysical property methods applied to fueled and un-fueled molten salts, Annals of Nuclear Energy, 146 (2020) 107608. https://doi.org/10.1016/j.anucene.2020.107608
Barnes J, Coutts R, Horne T, Thai J, Characterization of molten salt for application in molten salt reactors, PAM Review Energy Science and Technology, 6 (2019) 39–54. https://doi.org/10.5130/pamr.v6i0.1546
Mullin KM, Martin JH, Roper CS, Levi CG, et al., Transpiration cooling of a porous Nb-based alloy in high heat flux conditions, Inter. J. Thermal Sci., 196 (2024) 108758. https://doi.org/10.1016/j.ijthermalsci.2023.108758
Wood RJK, Lu P, Coatings and surface modification of alloys for tribo-corrosion applications, Coatings, 14(1) (2024) 99. https://doi.org/10.3390/coatings14010099
Ivanova A, Arkhipov P, Tkacheva O, Zaykov Yu, Experimental studies of the dynamic formation of the side ledge in an aluminum electrolysis cell, Russian Metallurgy (Metally), 2 (2020) 133–137. https://doi.org/10.1134/S0036029520020068
Tkacheva O, Arkhipov P, Kataev A, Rudenko A, et al., Solid phase formation during aluminium electrolysis, Electrochemistry Communications, 110 (2020) 106624. https://doi.org/10.1016/j.elecom.2019.106624
Isakov A, Chernyshev A, Apisarov A, Zaikov Yu, Electrodeposition of alloys from halide melts in solid state, Mater. Technol., 3 (2024) 20243036. https://doi.org/10.15826/elmattech.2024.3.036
Sato T, Ohashi K, Sudoh T, Haruna K, et al., Thermal expansion of a high purity synthetic diamond single crystal at low temperatures, Phys. Rev. B, 65 (2002) 092102. https://doi.org/10.1103/PhysRevB.65.092102
Bhaduri A. Mechanical Properties and Working of Metals and Alloys. Singapore: Springer Nature Singapore Pte Ltd.; 2018. 748 p. https://doi.org/10.1007/978-981-10-7209-3
Nedoseka A. Fundamentals of Evaluation and Diagnostics of Welded Structures. Cambridge, United Kingdom: Cambridge International Science Publishing Limited in association with Woodhead Publishing Limited; 2012. 639 p.
Mary TA, Evans JSO, Vogt T, Sleight AW, Negative thermal expansion from 0.3 to 1050 Kelvin in ZrW2O8, Science, 272 (1996) 90–92. https://doi.org/10.1126/science.272.5258.90
Cahn RW, The how and why of thermal contraction, Nature, 386 (1997) 22–23. https://doi.org/10.1038/386022b0
Dubey D, Mirhakimi AS, Elbestawi MA, Negative thermal expansion metamaterials: A review of design, fabrication, and applications, J. Manuf. Mater. Process., 8(1) (2024) 40. https://doi.org/10.3390/jmmp8010040
James JD, Spittle JA, Brown SGR, Evans RW, A review of measurement techniques for the thermal expansion coefficient of metals and alloys at elevated temperatures, Meas. Sci. Technol., 12 (2001) R1–R15. https://doi.org/10.1088/0957-0233/12/3/201
Ventura G, Risegari L. The art of cryogenics: low-temperature experimental techniques. Chapter 13. Measurements of thermal expansion. London, UK: Elsevier Ltd.; 2007. p. 289–296. https://doi.org/10.1016/B978-0-08-044479-6.X5001-8
Pathak PD, Vasavada NG, Thermal expansion of NaCI, KCI and CsBr by X–ray diffraction and the law of corresponding states, Acta Cryst., A26 (1970) 655–658. https://doi.org/10.1107/S0567739470001602
Hallam KR, Darnbrough JE, Paraskevoulakos C, Peter J, et al., Measurements by X-ray diffraction of the temperature dependence of lattice parameter and crystallite size for isostatically-pressed graphite, Carbon Trends, 4 (2021) 100071. https://doi.org/10.1016/j.cartre.2021.100071
Rao ASM, Narender K, Rao KGK, Krishna NG, Thermophysical properties of NaCl, NaBr and NaF by gamma–Ray Attenuation Technique, J. Modern Physics, 4 (2013) 208–214. http://dx.doi.org/10.4236/jmp.2013.42029
Flament C, Berthel B, Salvia M, at el. Characterization of the thermal behavior of a complex composite (clutch facing) combining digital image stereo correlation and numerical approach, Materials, 15 (2022) 2582–2605. https://doi.org/10.3390/ma15072582
Measurement of thermal expansion coefficient using strain gages. Micro-measurements. Vishay Precision Group, Tech. Note TN-513-1, Vishay Measurement Group, Document Number: 11063, (2010) 119–129.
Wallace G, Speer W, Ogren J, Es-Said OS, High-resolution methods for measuring the thermal expansion coefficient of aerospace materials, J. Mater. Engineering and Performance, 14 (2005) 563–564. https://doi.org/10.1361/105994905X64602
American Society for Testing and Materials. ASTM E289-2017. Standard Test Method for Linear Thermal Expansion of Rigid Solids with Interferometry (2017).
Cantor S, Metal dilatometer for determining density and expansivity of volatile liquids at elevated temperature, Rev. Sci. Instrum, 40 (1969) 967–968. https://doi.org/10.1063/1.1684132
Parker SS, Abdul‑Jabbar NM, Jackson JM, Monreal M, Feasibility of volumetric expansion of molten chlorides by conventional pushrod dilatometry, Radioanal. and Nucl. Chem., 331 (2022) 5259–5263. https://doi.org/10.1007/s10967-022-08641-2
Barron THK, Collins JG, White GK, Thermal expansion of solids at low temperatures, Advances in Physics, 29(4) (1980) 609–730. https://doi.org/10.1080/00018738000101426
Park J, Leong A, Zhang J, Density Measurements of Molten Salts, J. Chem. Eng. Data, 68(8) (2023) 1892–1898. https://doi.org/10.1021/acs.jced.3c00171
Vidrio P, Mastromarino S, Still E, et al., Density and thermal expansivity of molten 2LiF-BeF2 (FLiBe): measurements and uncertainty quantification, J. Chem. Eng. Data, 67 (2022) 3517–3533. https://doi.org/10.1021/acs.jced.2c00212
Parker SS, Long A, Lhermitte C, Vogel, et al. Thermophysical properties of liquid chlorides from 600 to 1600 K: Melt point, enthalpy of fusion, and volumetric expansion, J. Mol. Liq., 346 (2022) 118147. https://doi.org/10.1016/j.molliq.2021.118147
Cantor S, Ward WT, Moynihant CT, Viscosity and density in molten BeF2-LiF solutions, J. Chem. Phys., 50(7) (1969) 2874–2879. https://doi.org/10.1063/1.1671478
Yates B, Panter CH, Thermal expansion of alkali halides at low temperatures, Proc. Phys. Soc., 80 (1962) 373–382. https://doi.org/10.1088/0370-1328/80/2/304
Sanditov DS, Mantatov VV, Darmaev MV, Sanditov BD, O parametre Gryunayzena kristallov i stekol. [On the Gruneisen parameter of crystals and glasses], Techn. Phys. 79 (2009) 59–62. Russian.
Porter LJ, Justo JF, Yip S, The importance of Grüneisen parameters in developing interatomic potentials, J. Applied Physics, 82(11) (1997) 5378–5385. https://doi.org/10.1063/1.366305
Redmond AD, Yates B, The low temperature thermal expansion of thallous chloride and thallous bromide, J. Phys. C: Solid State Phys., 5 (1972) 1589–1603. https://doi.org/10.1088/0022-3719/5/13/009
Sunil K, Sharma BS, Thermoelastic properties of alkali halides at high temperatures, Indian journal of pure & applied physics, 50(6) (2012) 387–397.
Srivastava SK, Sinha P, Analysis of thermal expansion of NaCl and KCl crystals, Indian J. Phys., 85 (2011) 1257–1265. https://doi.org/10.1007/s12648-011-0151-2
Enck FD, Dommel JG, Behavior of the thermal expansion of NaCl at elevated temperatures, J. Applied Physics, 36(3) (1965) 839–844. https://doi.org/10.1063/1.1714229
Leadbetter AJ, Newsham DMT, Anharmonic effects in the thermodynamic properties of solids. A liquid gallium immersion dilatometer for the range 50–700 °C: thermal expansivities of Hg, Ga, NaCl and KCl, J. Phys. C: Solid State Physics, 2(2) (1969) 210–219. https://doi.org/10.1088/0022-3719/2/2/303
Pathak PD, Vasavada NG, Thermal expansion of LiF by X–ray diffraction and the temperature variation of its frequency spectrum, Acta Crystallographica, A28 (1972) 30–33. https://doi.org/10.1107/S0567739472000063
Ekinci Y, Toennies JP, Thermal expansion of the LiF(001) surface, Science, 563(1–3) (2004) 127–134. https://doi.org/10.1016/j.susc.2004.06.147
Sharma SS, Thermal expansion of crystals, Proc. Indian Acad. Sci. (Math. Sci.), 33 (1951) 283. https://doi.org/10.1007/BF03173262
Pathak PD, Pandya NV, Ghadiali MP, Thermal expansion of some alkali fluorides and magnesium oxide by X-ray diffraction, Indian J. Phys., 37 (1963) 293–298.
Panter CH, Thermal expansion of caesium bromide and potassium bromide, J. Phys. C: Solid State Physics, 7 (1974) 4483–4485. https://doi.org/10.1088/0022-3719/7/24/010
Anderson OL. Equation of state of solids for geophysics and ceramic science, New York, United States: Oxford University Press; 1995. 370 p. https://doi.org/10.1093/oso/9780195056068.001.0001
Smirnov MV, Khokhlov VA, Khokhlov VA, et al., Fiziko-khimicheskiye svoystva rasplavlennykh ftoridov shchelochnykh metallov [Physicochemical properties of molten alkali metal fluorides], J. Phys. Chemistry, 48 (1974) 467–469. Russian
Chrenková M, Danek V, Silný V, Density of the System LiF–KF–K2NbF7, Chem. Papers, 54 (2000) 272–276.
Abdullaev RN, Khairulin RA, Stankus SV, Density and thermal expansion of liquid salts LiF and LiF–NaF, Thermoph. and Aeromech., 30 (2023) 133–136. https://doi.org/10.1134/S0869864323010158
Janz GJ, Dampier FW, Lakshminarayanan GR, Lorenz PK, et al. Molten Salts: Volume 1, Electrical Conductance, Density, and Viscosity Data. Washington, USA: U.S. Government Printing Office; 1968. 139 p.
Minchenko VI, Stepanov VP. Ionnyye rasplavy: uprugiye i kaloricheskiye svoystva [Ionic melts: elastic and caloric properties]. Ekaterinburg: IHTE UB RAS; 2008. 367 p. Russian
Roberts RB, White GK, Thermal expansion of fluorites at high temperatures, J. Physics C: Solid State Physics, 19 (1986) 7167–7172. https://doi.org/10.1088/0022-3719/19/36/008
Rodnyi PA, Electron-hole and exciton processes in CaF2, SrF2, and BaF2 crystals (Review), Physics of the Solid State, 66 (2024) 155–171. https://doi.org/10.61011/PSS.2024.02.57911.215
Ekimova I, Minakova T, Ogneva T, Phisicochemistry of Alkaline-Earth Metals Oxides, Surface Advanced Materials in Technology and Construction, AIP Conf. Proc., 1698 (2016) 060014. http://dx.doi.org/10.1063/1.4937869
Ghalsasi P, Ghalsasi PS, Single crystal X-Ray structure of BeF2: alpha-quartz, Inorg. Chem, 50(1) (2011) 86–89. https://doi.org/10.1021/ic101248g
Wright AF, Fitch AF, Wright AC, The preparation and structure of the (alpha- and beta-quartz polymorphs of beryllium fluoride, J. Solid State Chem., 73(2) (1988) 298–304. https://doi.org/10.1016/0022-4596(88)90113-2
Gan CK, Al-Sharif AI, Al-Shormanb A, Qteish AA, First-principles investigation of the linear thermal expansion of BeF2: giant thermal expansion, RSC Adv., 12 (2022) 26588–26595. https://doi.org/10.1039/d2ra04860d
Takeda O, Yanagase K, Anbo Y, Aono M, et al., Density measurement of molten alkaline-earth fluorides using archimedean dual-sinker method, Int. J. Thermophys., 36 (2015) 2674–2686. https://doi.org/10.1007/s10765-015-1994-0
Kirshenbaum AD, Cahill JA, Stokes CS, The density of molten fluorides in the range of 1600–2500 K, J. Inorg. Nucl. Chem., 15(3–4) (1960) 297–304. https://doi.org/10.1016/0022-1902(60)80057-7
Krylosov AV, Polovov IB, Rebrin OI, Density and electrical conductivity of molten beryllium fluoride–alkali-metal chloride salt mixtures, Russian Metallurgy (Metally), 2 (2023) 229–234. https://doi.org/0.1134/S003602952302012X
Gadzhiev SM, Shabanov OM, Magomedova AO, Dzhamalova SA, Limiting electroconductivity and structure of molten chlorides of alkaline-earth metals, Russian J. Electrochem., 39 (2003) 1083–1088. https://doi.org/10.1023/A:1026171420132
Kim Y, Kang J, Viscosity of molten MgF2-LiF-MgO system and structure investigation using classical molecular dynamics simulations, J. Non-Crystalline Solids, 552 (2021) 120377. https://doi.org/10.1016/j.jnoncrysol.2020.120377
Li Z, Wu F, Theoretical insight into the structure of molten LiF, BF2, YF3 and ThF4, J. Radioanalytical and Nuclear Chem., 332 (2023) 1163–1170. https://doi.org/10.1007/s10967-023-08780-0
Janz GJ. Thermodynamic and Transport Properties for Molten Salts: Correlation Equations for Critically Evaluated Density, Surface Tension, Electrical Conductance, and Viscosity Data, Volume 17. Washington, USA: American Chemical Society and the American Institute of Physics; 1988. 309 p.
Roy S, Brehm M, Sharma S, Wu F, et al., Unraveling local structure of molten salts via X-ray scattering, raman spectroscopy, and Ab Initio molecular dynamics, J. Phys. Chem., 125B (2021) 5971–5982. https://doi.org/10.1021/acs.jpcb.1c03786
Bu M, Liang W, Lu G, Yu J, Static and dynamic ionic structure of molten CaCl2 via first-principles molecular dynamics simulations, Ionics, 27 (2021) 771–779. https://doi.org/10.1007/s11581-020-03852-7
Pavlatou EA, Papatheodorou GN, Raman spectroscopic study of in the crystalline, glassy and BeCl2 liquid states and of molten mixtures BeCl2–CsCl, Phys. Chem. Chem. Phys., 2 (2000) 1035–1043. https://doi.org/10.1039/A909120C
DOI: https://doi.org/10.15826/elmattech.2025.4.049
Copyright (c) 2025 Olga Yu. Tkacheva

This work is licensed under a Creative Commons Attribution 4.0 International License.

